Over the course of the forecast period, the market for viral vector and plasmid DNA testing services is anticipated to grow at a CAGR of 25.0%, owing to rising business expansion through the opening of facility centres, mergers and acquisitions, product launches, and other activities by market players. For instance, Merck KGaA stated in May 2016 that it was expanding its Carlsbad, California facility's operational capability to provide cell and gene therapy services. Moreover, the facility aids in the creation of items for use on a clinical and commercial scale.
The Viral Vector and Plasmid DNA Testing Services Market is a rapidly growing industry that serves the needs of the pharmaceutical, biotech, and academic research communities. Viral vectors and plasmid DNA are essential tools for gene therapy, vaccine development, and gene editing, among other applications. These vectors and DNA are widely used in the development of gene therapies, which are emerging as a promising approach for the treatment of a range of diseases, including cancer, genetic disorders, and viral infections.
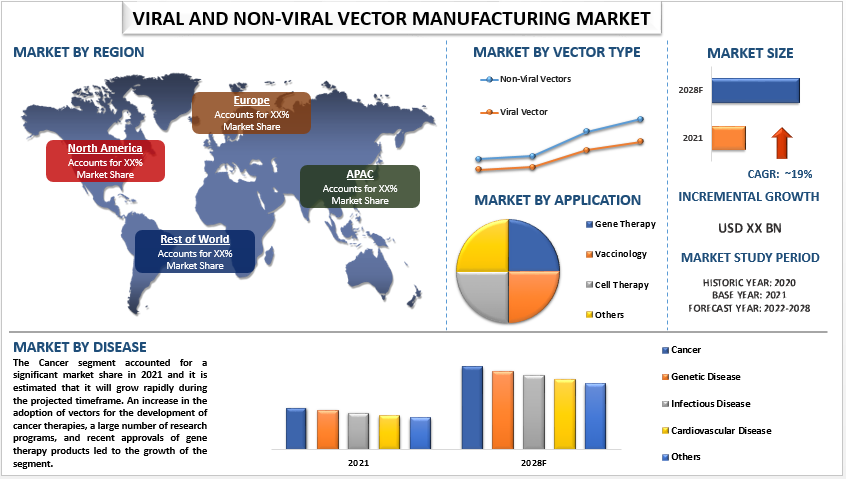
Due to the rising number of new products or services introduced by the manufacturers, which is anticipated to fuel market expansion, the safety segment had a dominating position among testing services in the viral vector and plasmid DNA testing services market in 2018.
Viral vectors and plasmid DNA are widely used in gene therapy. Gene therapy involves the delivery of a functional gene to replace a defective gene or to introduce a new gene into the body. This approach has the potential to cure genetic disorders that have no effective treatments. Viral vectors are often used for gene therapy, as they are highly efficient in delivering genes to target cells. However, viral vectors have limitations, such as the risk of immune responses and potential toxicity. Plasmid DNA is an alternative vector that can be used for gene therapy. Plasmid DNA is safe, non-infectious, and can be easily manufactured. Plasmid DNA is also a preferred vector for DNA vaccines, which are used to stimulate an immune response against infectious agents.
Read More:
https://knackersblogger.blogspot.com/2023/03/viral-vector-and-plasmid-dna-testing.html




Industry Overview of the Report 2026:The market intelligence report on the Global Viral Vector and Plasmid DNA Manufacturing Market offers the readers a 360° market overview with definitions, market segments, applications, raw material used, product details, cost structures, production processes, and other essential data.
The study evaluates the global market landscape, with an in-depth analysis of product pricing, production and consumption volume, cost, value, production capacity, supply and demand dynamics, annual market growth rate, and market estimation till 2026.This report covers the recent COVID-19 incidence and its impact on Viral Vector and Plasmid DNA Manufacturing Market.
The pandemic has widely affected the economic scenario.
This study assesses the current landscape of the ever-evolving business sector and the present and future effects of COVID-19 on the market.To get a sample of this report, with 30 minutes free consultation, visit @ https://www.reportsanddata.com/sample-enquiry-form/1938 The Global Viral Vector and Plasmid DNA Manufacturing Market Can Be Segmented By:Leading Players: FUJIFILM Diosynth Biotechnologies, Kaneka Eurogentec S.A, Fin vector, Spark therapeutics, Cobra biologics, Cell and gene therapy catapult, Renova therapeutics, Shenzhen SiBiono GeneTech Co., Ltd, Thermo Fisher Scientific, Inc, 4D Molecular Therapeutics are the major players in the Viral vector and plasmid DNA manufacturing market.Segmentation: By Product (Revenue, USD Million; 2016-2026)Plasmid DNAViral VectorsNon-viral VectorsBy Workflow (Revenue, USD Million, 2014 - 2026)Upstream ProcessingVector Amplification & ExpansionVector Recovery/HarvestingDownstream ProcessingPurificationFill-finishBy Disease (Revenue, USD Million; 2016-2026)CancerInherited DisordersViral InfectionsOthersBy Application (Revenue, USD Million, 2016-2026)Antisense, RNAi, & Molecular TherapyVaccinologyBy Sales channel (Revenue, USD Million; 2016-2026)Channel salesDirect SalesBy End user: (Revenue, USD Million; 2016-2026)Pharmaceutical and Biopharmaceutical CompaniesResearch InstitutesRegional Viral Vector and Plasmid DNA Manufacturing Market (Regional Output, Demand & Forecast by Countries):- North America (United States, Canada, Mexico)South America (Brazil, Argentina, Ecuador, Chile)Asia Pacific (China, Japan, India, Korea)Europe (Germany, UK, France, Italy)Middle East Africa (Egypt, Turkey, Saudi Arabia, Iran) and More.
The study also provides a meticulous evaluation of the key threats faced by the pioneers of the market, which allows the participants to comprehend the challenges they may encounter in the future as part of the global market in the forecast duration.The Market Report Contains The Following Chapters:Chapter 1: This report on the Global Viral Vector and Plasmid DNA Manufacturing Market brings in one place all the vital information pertaining to the sector.Chapter 2: The report comprises of a detailed analysis of players that account for a significant portion of the global market share in the Viral Vector and Plasmid DNA Manufacturing industry, highlighting the company’s latest technological advancement in the market, and the product profile currently available in the market, as well as the regions where they predominantly operate.Chapter 3: It helps understand the major product segments and the future of the Global Viral Vector and Plasmid DNA Manufacturing Market.
It gives strategic measures in key business segments based on market estimations.Chapter 4: The report also provides an eight-year forecast survey predicting the growth of the market in the forecast duration.The Viral Vector and Plasmid DNA Manufacturing industry research report outlines aspects like production, demand and supply, sales, and the contemporary market scenario exhaustively.

Transparency Market Research (TMR) has published a new report titled, ‘Viral Vector & Plasmid DNA Manufacturing Market - Global Industry Analysis, Size, Share, Growth, Trends, and Forecast, 2019–2027.’ According to the report, the global viral vector & plasmid DNA manufacturing market was valued at US$ 0.4 Bn in 2018 and is projected to expand at a CAGR of 23.3% from 2019 to 2027.
Viral vectors are generally utilized as vectors for gene therapy.There are various types of vectors, such as lentivirus, adenovirus, adeno-associated virus (AAV), and others, which include herpes simplex virus and chimeric viral vectors.
Plasmid DNA is used as raw material in the production of various viral vectors.Technological advancements, increase in awareness about viral vector-based treatments, and rise in demand for vector-based products for research are the major factors anticipated to drive the global viral vector & plasmid DNA manufacturing market during the forecast periodNorth America held major share of the global viral vector & plasmid DNA manufacturing market in 2018.
Availability of the U.S. FDA (Food and Drug Administration) approved gene therapy products and rich product pipeline are the factors expected to augment the viral vector & plasmid DNA manufacturing market in the region during the forecast period.The viral vector & plasmid DNA manufacturing market in Asia Pacific is likely to expand at a high CAGR of5%from 2019 to 2027, due to recent developments in regenerative medicines, rise in prevalence of chronic diseases, and increase in demand for gene therapies.
Additionally, demand for viral vector & plasmid DNA is increasing in emerging markets such as India and China.
Request COVID Analysis on Viral Vector & Plasmid DNA Manufacturing Market – https://www.transparencymarketresearch.com/sample/sample.php?flag=covid19_id=30428Rise in Prevalence of Cancer, Genetic Diseases, and Infectious Diseases to Propel Global Viral Vector & Plasmid DNA Manufacturing MarketRise in prevalence of cancer and genetic diseases is projected to augment the global viral vector & plasmid DNA manufacturing market during the forecast periodAccording to the Cancer Research UK, 17 millionnew cases of cancer were reported globally in 2018 and the four most common types of cancer were breast, lung, bowel, and prostate, which account for around 43% of all new casesAccording to the National Cancer Institute, cancer is among the leading causes of death across the world, and the number of new cases per year is anticipated to rise to 6 millionby 2030.Request Customization on Viral Vector & Plasmid DNA Manufacturing Market Report – https://www.transparencymarketresearch.com/sample/sample.php?flag=CR_id=30428Plasmid DNA Segment Dominated Global Viral Vector & Plasmid DNA Manufacturing MarketIn terms of type, the global viral vector & plasmid DNA manufacturing market can be classified into lentivirus, adenovirus, adeno-associated virus (AAV), plasmid DNA, and othersThe plasmid DNA segment dominated the global viral vector & plasmid DNA manufacturing market in 2018and the trend is expected to continue during the forecast periodIncrease in use of plasmid DNA as raw material in the manufacture of viral vectors is likely to drive the segment during the forecast periodCancer to be Promising Segment of Global Viral Vector & Plasmid DNA Manufacturing MarketBased on disease, the global viral vector & plasmid DNA manufacturing market has been categorized into genetic disorders, cancer, infectious diseases, and othersThe cancer segment dominated the global viral vector & plasmid DNA manufacturing market in 2018and the trend is projected to continue during the forecast period owing to large number of ongoing clinical trials and availability of approved viral vector-based cancer drugsBiotech Companies to be Lucrative Segment of Global Viral Vector & Plasmid DNA Manufacturing MarketIn terms of end-user, the global viral vector & plasmid DNA manufacturing market has been categorized into biotech companies and research institutesThe biotech companies segment held major share of the global viral vector & plasmid DNA manufacturing market in 2018Increase in number of viral vector-based cancer drugs being manufactured by multinational players and several clinical trials underway for the treatment of various cancers are anticipated to propel the segment during the forecast periodBuy Viral Vector & Plasmid DNA Manufacturing Market Report – https://www.transparencymarketresearch.com/checkout.php?rep_id=30428=SNorth America to Dominate Global Market; Asia Pacific to Offer Significant Incremental OpportunityIn terms of region, the global viral vector & plasmid DNA manufacturing market has been segmented into North America, Europe, Latin America, Asia Pacific, and Middle East & AfricaNorth America is projected to account for major share of the global viral vector & plasmid DNA manufacturing market during the forecast period, owing to high acceptance of viral vector-based gene therapies for the treatment of rare diseases and availability of FDA-approved gene therapiesThe viral vector & plasmid DNA manufacturing market in Asia Pacific is anticipated to grow at a rapid pace in the next few years.